Since I started in the lab, I had the impression that the SB buffer we use for gel electrophoresis did not work quite as well as the more commonly used TAE buffer, but as I wasn’t running many gels I was too lazy to run a test. Also, Wikipedia said I was wrong, and who am I to doubt Wikipedia?
The supposed advantage of SB is that you can run gels at higher voltage (and therefore, one would expect, faster) while still getting sharp bands and not having your gel overheating and melting in the electrophoresis chamber. Still, my gels seem to run forever even at 200V, and they look kinda shitty.
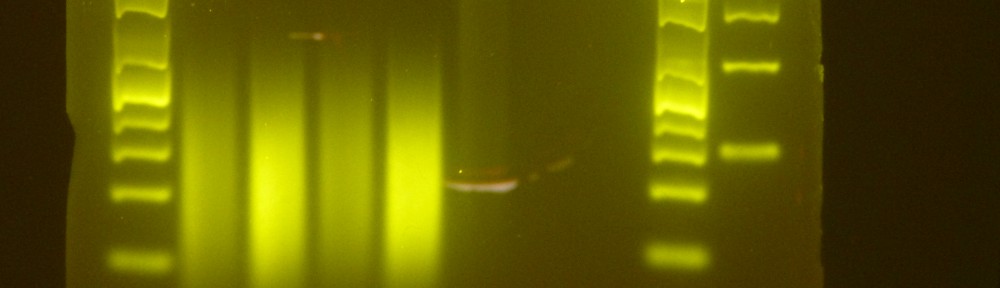
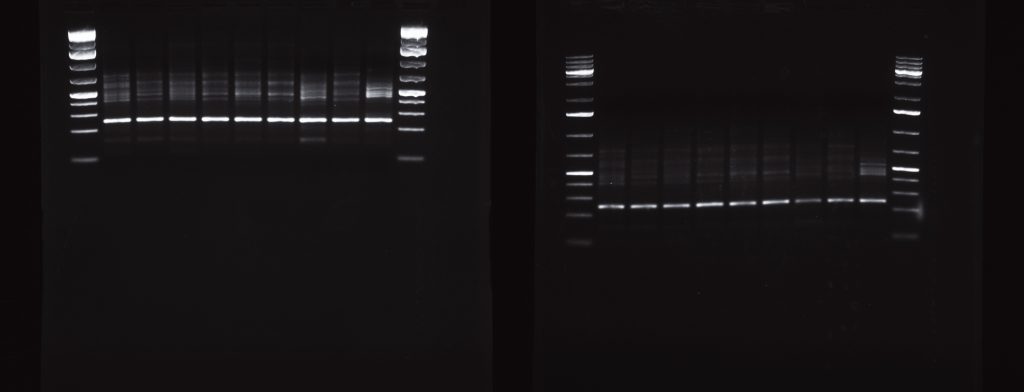
So I had my new work-learn student Jasmine to prepare also some 50X TAE. I then split some test PCRs I had lying around in two, and load them into two 1.5% agarose gels, one made with SB buffer and one with TAE buffer. I run them at the same time attached to the same power source, at 400 mA and 120V. The following picture was taken after about 1 hour. The SB gels is on the left, the TAE one is on the right.
As you can see the gel with SB buffer runs much slower than the TAE one at the same voltage. So, yes, you can probably run an SB gels at higher voltages (I’ll assume it is true that SB gels can withstand higher currents without overheating), but you’ll get comparable result running a TAE gel at lower voltages (minus the wasted electricity).
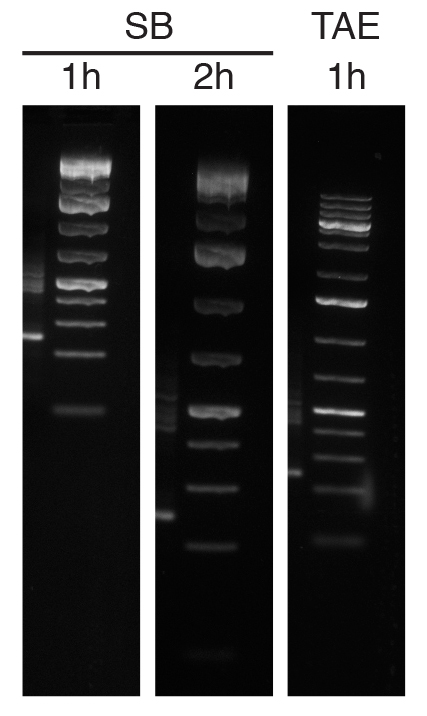
If that would be the only difference, while annoying, it would hardly be sufficient reason to change the lab buffer of choice. The real problem is that the bands do not look so good on SB. You can already see it in the two pictures in the figure above, but to make a better comparison I run the SB gel for another hour, until it looked more similar to the 1-hour TAE gel. The following figure is a comparison between the ladder in a SB gel run for 1 or 2 hours and a TAE gel run for 1 hour. Again, these are all 1.5% gels run at 120V.
As you can hopefully see, starting from 500 bp up (the fifth band from the bottom) bands start to loose sharpness in the SB gels, and the larger bands (4Kb up) are a single blurry blob. By contrast, all bands are sharp and well defined in the TAE gel.
Based on the evidences, I put forward a motion to dismiss SB as the standard electrophoresis buffer in the lab and switch to TAE instead (I will certainly do so myself, but it would be messy to have two electrophoresis buffers going on at the same time). I am happy to (have Jasmine) make a couple of liters of 50X TAE, which should last us several months.
As a side note, I realized that the vague Street Fighter 2 reference in the title is probably lost on most members of the lab. Alas, that’s how old I am.