Research Program
General Introduction: My long-term goal is to understand the evolution of sexual reproduction. For many non-biologists the biggest barrier to understanding this field is accepting that , for biologists, sex poses a serious problem . We all know how sexual reproduction works: each person has two copies of each chromosome (and therefore two complete sets of genes) in each of their normal cells, but produces eggs or sperm by a special cell division called meiosis which gives these germ cells only one copy of each chromosome (one complete set of genes). When the egg and sperm fuse, their two sets are combined, so that each child gets two copies of each chromosome, one from each parent, and has new combinations of its parents' characteristics. Why such a complicated mode of reproduction evolved is one of the biggest unsolved problems in biology. The difficulty is not so much in understanding why there are males and females, but in understanding why most organisms (plants, animals, and many fungi and single-celled protists) give their progeny only half of their genes, rather than producing offspring that are exact copies of themselves. Old-fashioned textbooks explained sex as being good for the long-term evolution of the species; pointing out that offspring that differed from their parents might sometimes be better adapted, especially if the environment has changed. But sexual reproduction is difficult and expensive, and most biologists now believe that any advantage to the species would be too unpredictable, too weak, and too far in the future to make up for the immediate problems that sexual reproduction causes for individuals. Thus sex poses a serious paradox for biologists-its ubiquity implies that it must have major and reliable benefits, but these remain unresolved.
Research Objectives
The three components of my research program are described below. I began my research career with the broad goal of understanding the evolution of sexual reproduction, and more specifically of using investigations into the molecular biology underlying sexual processes to shed light on their evolutionary causes. I was diverted from my original plan to study true sexual reproduction in eukaryotes by the need to first critically test the widely-held assumption that bacterial competence evolved for genetic exchange (Part I). As our research confirms that this assumption is indeed false, we have now proceeded to the next steps, investigating sexual reproduction in the most divergent groups of eukaryotes (Part II), and analyzing problems posed by specific components of sexual reproduction, in several systems (Part III).
The first step in my research program was to test the widely-held assumption that bacterial competence is a form of sex. Many groups of bacteria have such natural competence systems, which enable them to take up DNA from their environments. Because the DNA may then recombine with the cells' chromosomes, most researchers have assumed that competence evolved for genetic exchange, and some have used it as a model system for investigating the evolution of sex. Our analysis of competence in Haemophilus influenzae instead has revealed that competence has a primarily nutritional function, with the DNA serving mainly as a source of nucleotides. Thus competence produces genetic exchange only as a side effect, and has little in common with sex.
Experimental work: Our research to date has demonstrated the following:
--DNA damage does not induce competence in H. influenzae or Bacillus subtilis, contrary to predictions of the competing DNA-repair hypothesis. [Publication #9 (included)]
--Induction of competence in H. influenzae is controlled by adenylate cyclase and a fructose-specific phosphotransferase system, which together provide signals connecting shortages of preferred catabolites to induction of operons allowing use of alternative energy resources [Publications #3 (included) and #8; Irene Dorocicz's M.Sc. project, Leah Macfadyen's Ph.D. project, Shaun Cordes' project. Collaborators' contributions to publication #3: J. Reizer in M. Saier's lab provided the data in Tables 2 and 3.].
--H. influenzae is highly specialized for use of its mucosal environment, which contains substantial concentrations of host DNA that could provide H. influenzae with nucleotides [Publication #19].
--Competence induction in H. influenzae is also controlled by the sxy gene [Publications #6 (included) and #10]. The secondary structure of the sxy mRNA may serve to permit sxy expression only when nucleotide pools are depleted [Laura Bannister's Ph.D. project].
--Other experiments in progress address the role of the cAMP phosphodiesterase in regulation of competence, and the modes of action of several new mutations that cause extreme hypercompetence. I also plan to recruit a Ph.D. student (supported by our MRC Studentship) to investigate the role of uptake sequences in competence.
Theoretical work: In addition to the experimental work, I have used theoretical analysis, primarily computer simulations, to show that competence is unlikely to provide the same recombination benefits as meiotic sex. This is due to two critical differences. First, the available DNA will carry an excess of deleterious mutations, and second, it may carry alleles that preclude competence, especially if the competent cell has recently arisen in a population of non-competent cells [Publications #2 (included) and #14. Collaborators' contributions to publication #2: I posed the scientific questions, Dr. Dean did most of the mathematical analysis, and Dr. Schrag provided advanced mathematical skills. Dr. Dean and I shared equally in writing the paper. Publication #14 was produced while I was a post-doctoral fellow with R. Lewontin.].
I have recently been awarded a 5-year MRC research grant (ranked first of 40 applications in its field) and an accompanying MRC-funded Ph.D. Studentship to continue our work on H. influenzae. [A copy of the MRC proposal is available at (http://www.zoology.ubc.ca/~redfield/research.html).] By the end of this award I expect to have demonstrated that competence development in H. influenzae is indeed triggered by depletion of nucleotide pools. I believe that such a finding, combined with what we have already learned about the regulation of DNA uptake by energy limitation and about the abundance of unrelated DNA in mucosal environments, would show conclusively that competence exists primarily because of its nutritional consequences. As the other 'parasexual processes' of bacteria are already known to have non-sexual causes, this demonstration would overturn the widely-held assumption that bacteria are sexual organisms.
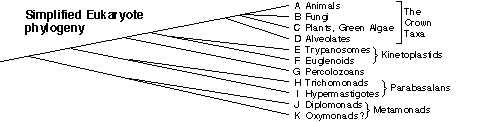
My goals here are to determine when true sexual reproduction arose in eukaryotes, and how the most deeply divergent forms of sexual reproduction differ from those we know. To date most research into the mechanisms and evolution of sex has been carried out in animals, fungi and plants, which form a closely-related group at the crown of the eukaryotic evolutionary tree shown below. However true sexual reproduction arose very early in the eukaryotes, possibly predating the deepest known branch point in this tree. Consequently one component of my research aims to characterize sexual reproduction in the organisms that are most divergent from the crown taxa.

When did sexual reproduction arise?
Phylogenetic placement and sexuality of the early-branching Hypermastigote and Oxymonad protists. These are thought to be the most ancient eukaryotic lineages capable of sexual reproduction. However both their phylogenetic placement and their sexuality remain uncertain, partly because the organisms cannot be maintained in culture. We are using PCR and fluorescence in situ hybridization to obtain DNA sequences for phylogenetic analysis [Manuscript submitted, Joel Dacks' M.Sc. project]. This has confirmed the deep divergence of the Hypermastigotes. We are currently analyzing Oxymonad sequences, and will develop molecular methods to examine these organism's genomes for sex-related genes.
How does sexual reproduction take place?
Sexual reproduction in the amoeboflagellate Naegleria. Naegleria are deeply branching but easily cultured, and genetic tools are being developed. Although sexuality has not been observed in lab cultures, the population genetics implies that at least some species of Naegleria freely recombine in nature. We will use population and molecular genetics methods to investigate the mechanism of genetic exchange. We have provided John Logsdon at Dalhousie with N. gruberi DNA to construct a library, and we plan to jointly investigate the presence and activity of sex-related genes.
III. Specific problems of sexual reproduction:
Male mutation rates and the cost of sex for females: This work combined well-established analysis of the costs and benefits of sex for females with a recent demonstration that most deleterious mutations arise in the male germ line, and concluded that the combined costs for females would exceed the benefits under most if not all reasonable assumptions [Publication #7 (included); this research received a great deal of attention, including an accompanying News and Views article in Nature, a full page article in the New York Times, and a joke by Jay Leno on the Tonight Show.].
Evolution of the mechanism of meiotic recombination: We have published a theoretical analysis of meiotic crossing over, showing that the presently-understood break-repair mechanism is untenable because the initiating hotspots are destroyed by gene conversion [Publication #1 (included), Alan Boulton's Ph.D. project]. The break-repair mechanism has been widely accepted, so the implications of our analysis are substantial. In work in progress, we are extending this analysis to a comprehensive model incorporating haploidy, facultative sexual reproduction, and recombination interference between multiple hotspots [Aliza Israel's theoretical project]. We are also preparing to test the predictions of our analysis experimentally in the yeast Saccharomyces cerevisiae, by following the frequencies of active and inactive hotspot alleles through many sexual generations [Aliza Israel's experimental project].
Plans for the future:
Sexual reproduction is one of the critical factors distinguishing eukaryotes from bacteria and archaea, and the genetic exchange sex causes is the foundation of eukaryotic genetic analysis. Our inability to explain why sex exists is an embarassing gap in our understanding of evolutionary processes. I believe that this can best be changed by critically examining the molecular and evolutionary foundations of sex, using the commonalities between sex in the crown taxa and in our most-distant sexual relatives to determine which features are truly primitive, and the molecular mechanisms and consequences to indicate the evolutionary forces responsible for their success. Thus my main experimental goal will continue to be characterization of the precursors and most primitive examples of meiotic recombination and syngamy in the earliest-branching eukaryotes. Theoretical approaches will concentrate on the points of disagreement between experimental observations and theoretical predictions about the molecular biology of sex.