Research
My laboratory studies one of the fundamental molecular trafficking pathways within the cell:
bidirectional transport of macromolecules between the nucleus and the cytoplasm.
To investigate this problem we use cellular and molecular techniques in combination with
fluorescence and electron microscopy. We also explore possible practical applications by
incorporating viral research into our studies. We study how viruses deliver their genome to
the cell nucleus. These studies on nuclear import of viral genomes are of particular importance
because they may lead to the development of treatments and drugs that block nuclear uptake of viruses
and thereby viral replication and propagation of infections. The viruses presently under
investigation in our lab include Influenza virus, Hepatitis B virus and parvoviruses.
Molecular trafficking between the nucleus and the cytoplasm occurs through specialized
multi-protein channels called nuclear pore complexes (NPCs). As the NPC is the major player
in nucleocytoplasmic transport, we are also molecularly and structurally characterizing the NPC.
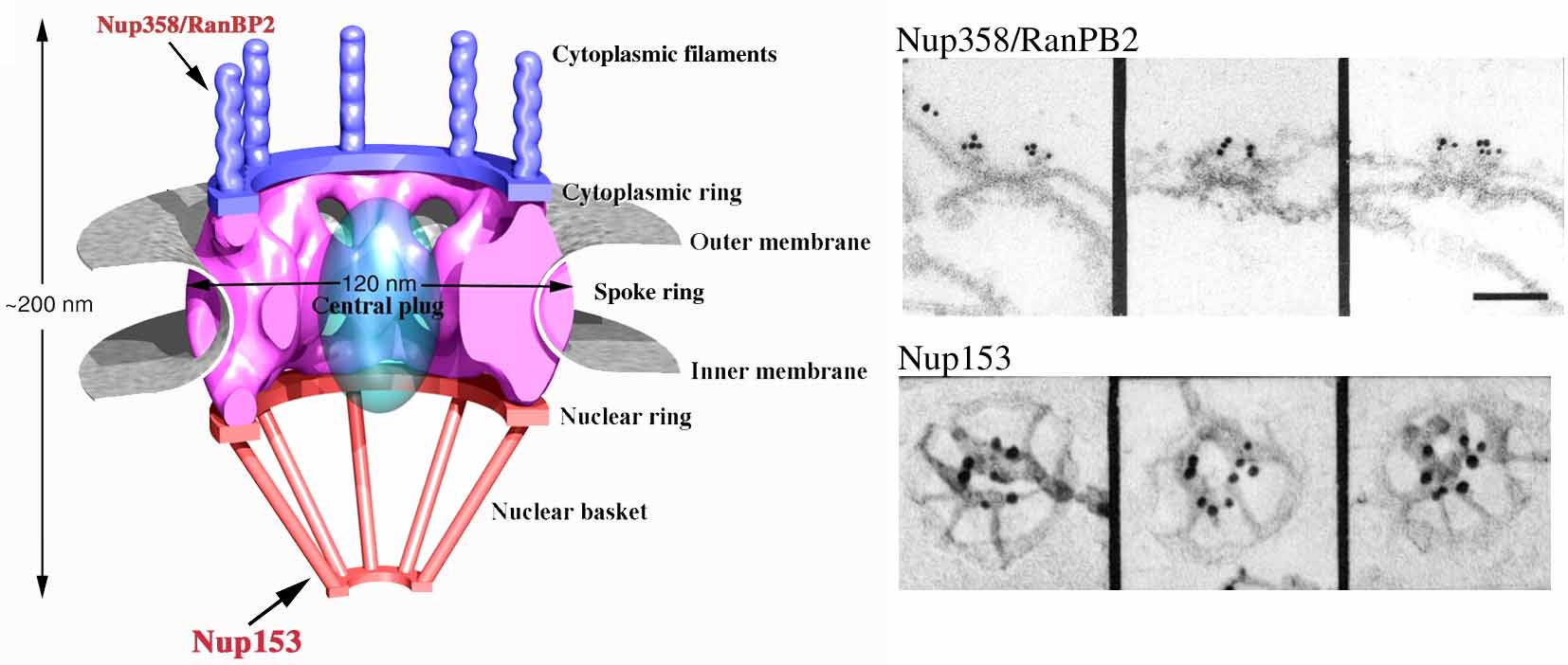
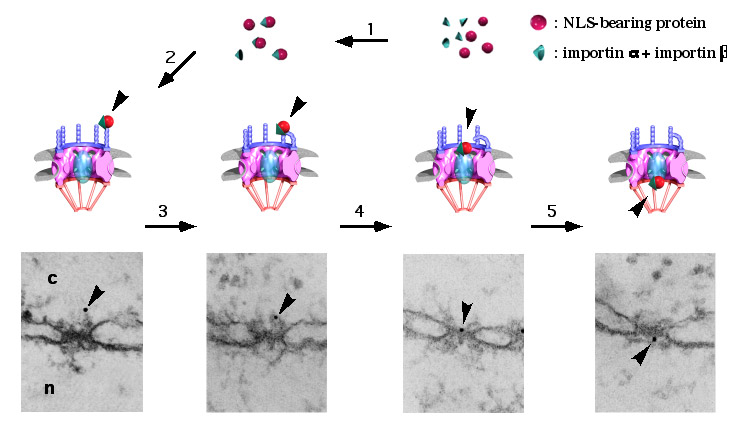
To study nuclear import in vivo we use a model system consisting on microinjection of transport substrates into the cytoplasm of Xenopus oocytes in combination with colloidal gold labeling and high-resolution electron microscopy.
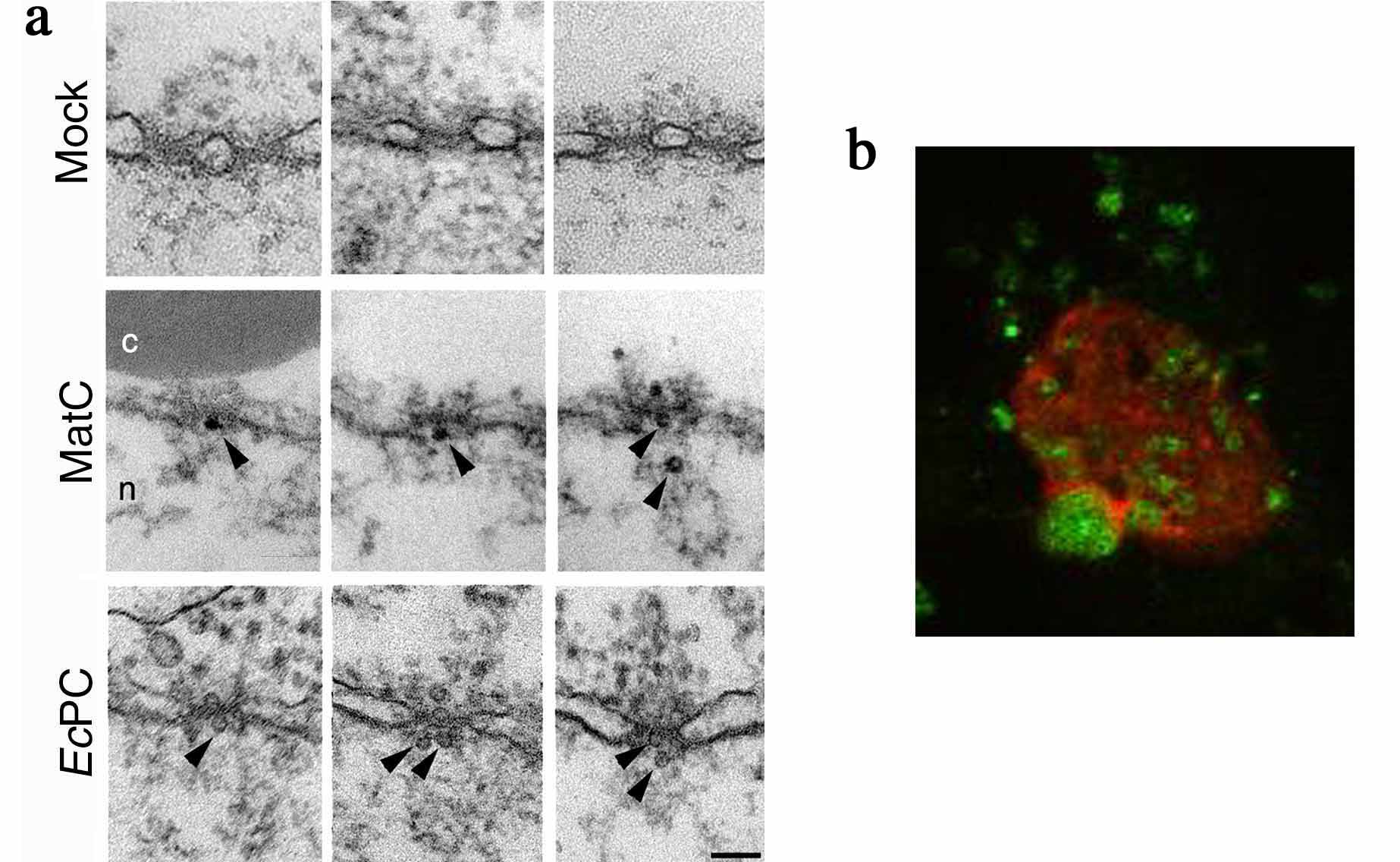
For the studies of nuclear import of viruses we also use the Xenopus system, as well as cells infected with virus followed by visualization using indirect immuno-localization and fluorescence microscopy.