Chapters
Lodish 4th edition: Chapter 21 pages 921 - 924
Moyes: Chapter 3.
ATPase pumps
ATPase pumps use the energy from ATP to transport ions against their
concentration gradients.
A lot of energy in the cell (25% of the ATP) is used up by the ATPase pumps.
Used for many different ions. Essential to maintain the Na+, K+ and Ca+2
concentration gradients that we will be talking about when we discuss
cotransport, action potentials, and muscle contraction.
We will discuss mostly P-class pumps a class of ATPase pump that is
phosphorylated to drive transport. There are a number of other classes
diagrammed above and listed in Table 15-2 in your text book.
P-class pumps
Na+/K+ ATPase pump
pumps 2 K+ in and 3 Na+ out
- important for many cellular functions (osmotic balance of cells)
- 3 Na+ out for every 2 K+ ions in
- uses ATP as energy source
- binding of phosphate from ATP drives conformation change that allows ions to
be transported to appropriate sides
- an asparate residue becomes phosphorylated and the energy transfer changes
the proteins conformational shape
- Na+ binding sites switch from high affinity on inside to low affinity on
outside to allow for binding of Na+ on inside and release of Na+ ions on
outside.
- K+ binding sites switch from high affinity on outside to low affinity on
inside for the same reason
- can be blocked with poisons like ouabain or digitalis
- blocked pump can't transport ions across membrane and the chemical gradients
for Na and K slowly disappear due to continuous action of the leak channels,
resting potential removed.
- the potential built up in the Na+ ions will be used by many different
processes i.e. cotransporters, neuronal signaling etc.
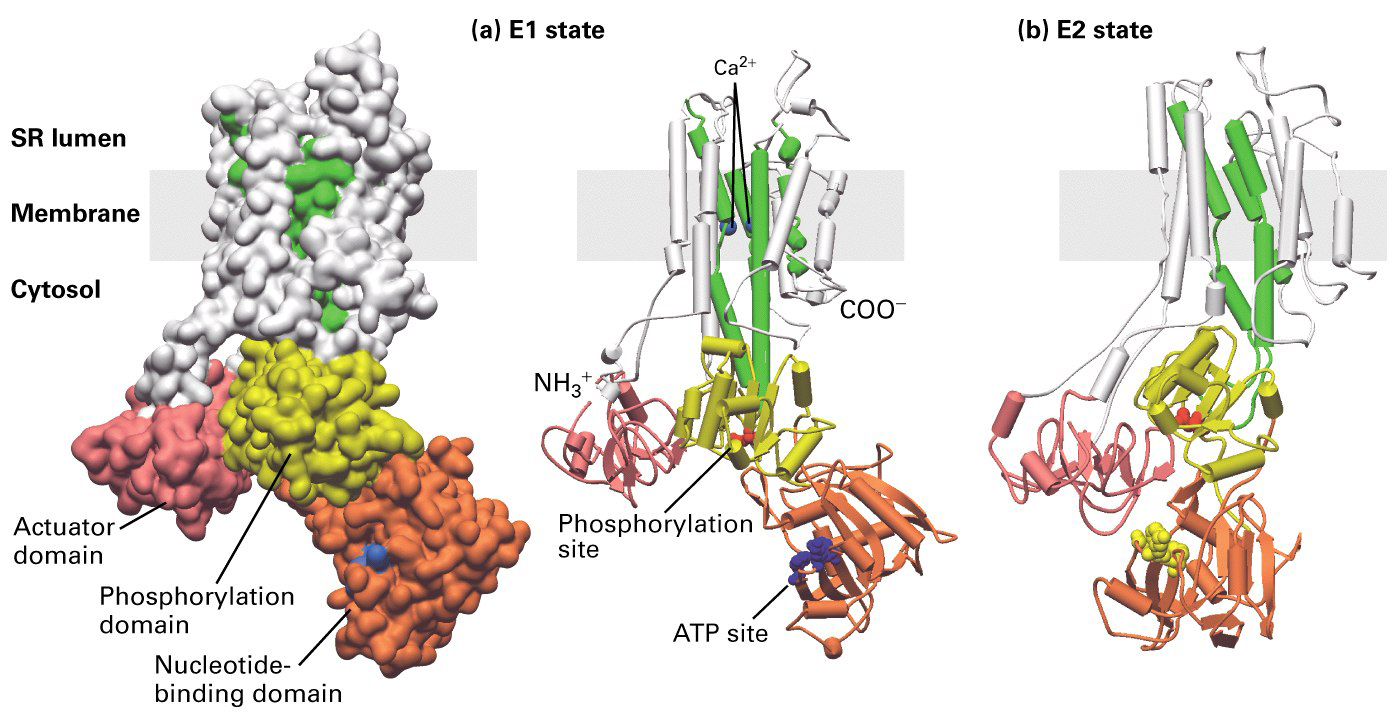
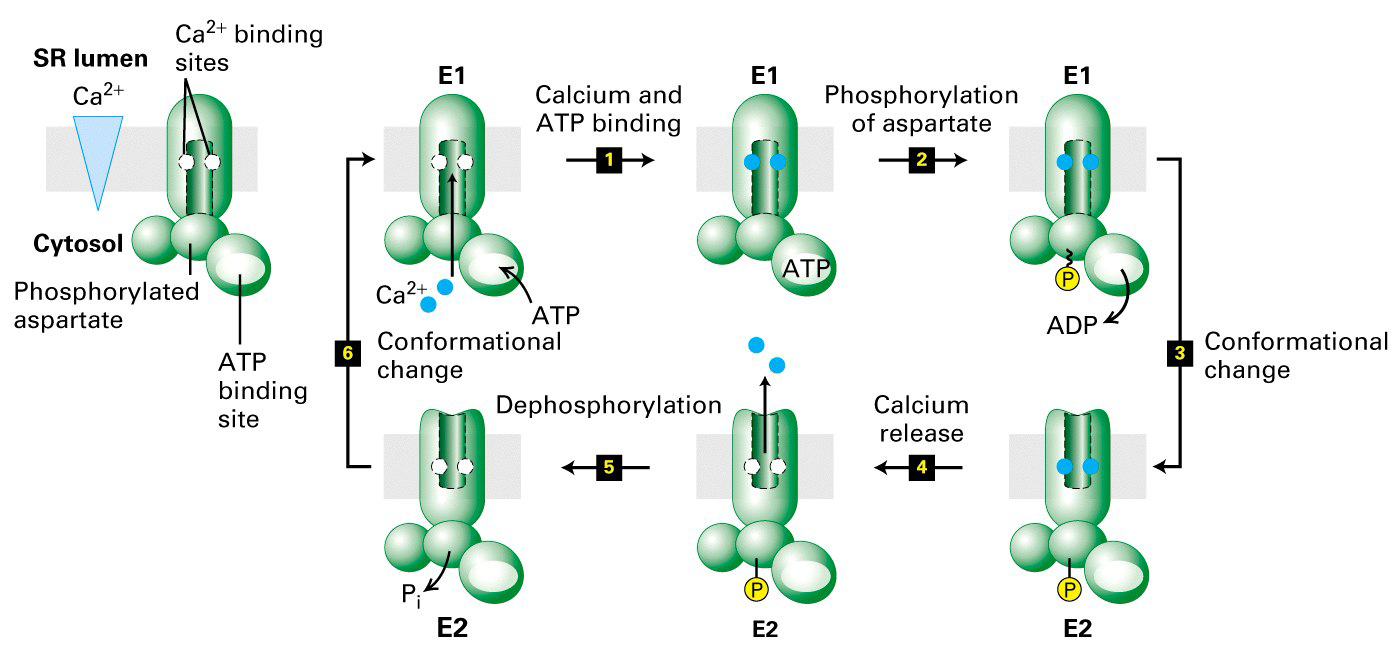
Ca+2 ATPase pump
- pumps 2 Ca+2 ions out for every 1 ATP molecule used
- again uses ATP to drive Ca+2 out against a very large concentration gradient
- internal Ca+2 binding sites have a very high affinity (in order to overcome
extremely low Ca+2 concentrations inside cell)
- energy transfer from ATP to the aspartate of the Ca+ ATPase causes a protein
conformational change and Ca+2 transported across membrane
- Ca+2 binding sites on outside are low affinity and Ca+2 is released
- the transfer of eneragy from the ATP to the pump triggers a conformational
change that moves the protein and allows the translocation of Ca+2 across the
membrane. At the same time the Ca+2 binding sites change from high to low
affinity.
- in muscle cells the Ca+2 ATPase is the major protein found in the membrane
of the sacrcoplasmic reticulum (SR). As we will see the SR is a storage site
for Ca+2 that is release to drive muscle contraction. The Ca+2 ATPase will
remove excess Ca+2 from the cytoplasm and pump it into the lumen of the SR.
80% of the protein in the SR is the Ca+2 ATPase. Therefore this made it easy
to purify and study in liposomes as we discussed in class.
Figure 15-12 is a schematic diagram of a potential model of the Ca+2 ATPase
pump.
- 10 transmembrane regions are shown (these are predicted based on an
alpha-helical like structure and perhaps hydrophobic properties, - those
helices that interact with the Ca+2 of course will not be so hydrophobic)
- the large intracellular loops were thought to be important for pump
function.
How could you go about testing which regions are important for function?
One way possible way is to carry out "structure/function" experiments.
- this is possible if you have cloned the pump (or the protein you are
interested in)
- to test its function you can make mRNA from the full-length clone in the
test tube and then express the mRNA by injecting into a cell or translating in
an in vitro system
- the cell could be a tissue culture cell or a Xenopus oocyte for instance if
the protein is not normally expressed in these membranes
- or the protein could be translated and introduced into liposomes which are
"balls" or spheres of pure lipids (outlined in Figure 15-4 below).
- once the protein is is the membrane you can now test the function of the
expressed protein
- using a Ca+2 tracer, radioactive Ca+2 or a Ca+2 sensitive dye for instance
you can now test the function of the pump (some researchers follow the current
generated by the Ca+2 crossing the membrane)
- the next step is to make changes in the pump by altering the DNA (remove
regions that you think are important by deleting or changing their coding
sequence) and then express the mRNA from these mutant pumps, inject this into
your cells or liposomes and test the function of the pump
- in this way you can define the regions that are important, like the ATP
binding region, the asparatate, and the energy transduction regions outlined
in the figure above.
V-class pumps
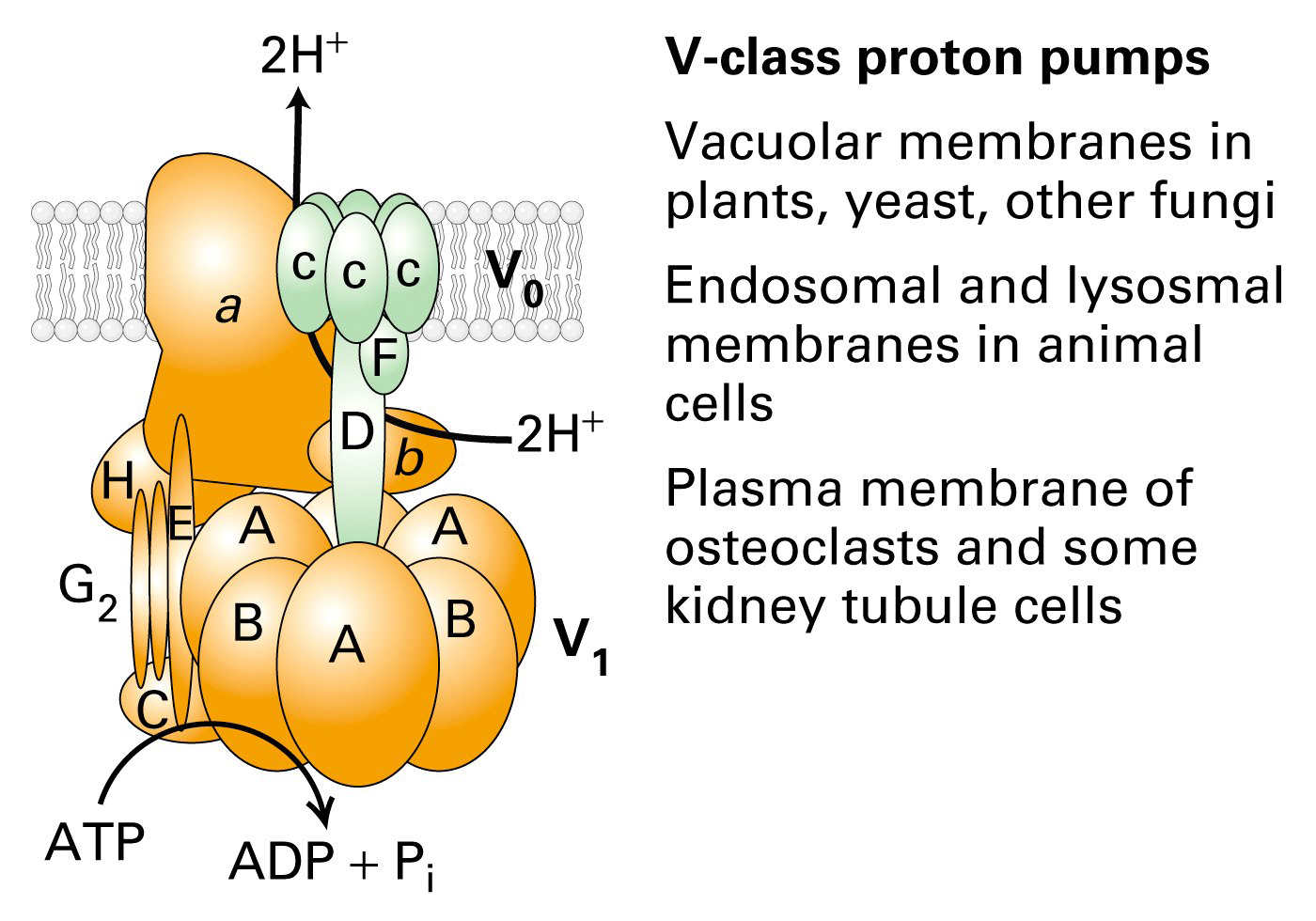
These pumps transport H+ only.
- found in lysosomes, endosomes and plant vacuoles.
- transport H+ ions to make the lumen or inside of the lysosome acidic (pH 4.5
- 5.0)
- many of these pumps are paired with Cl- channels to offset the electrical
gradient that is produced by pumping H+ across the membrane.
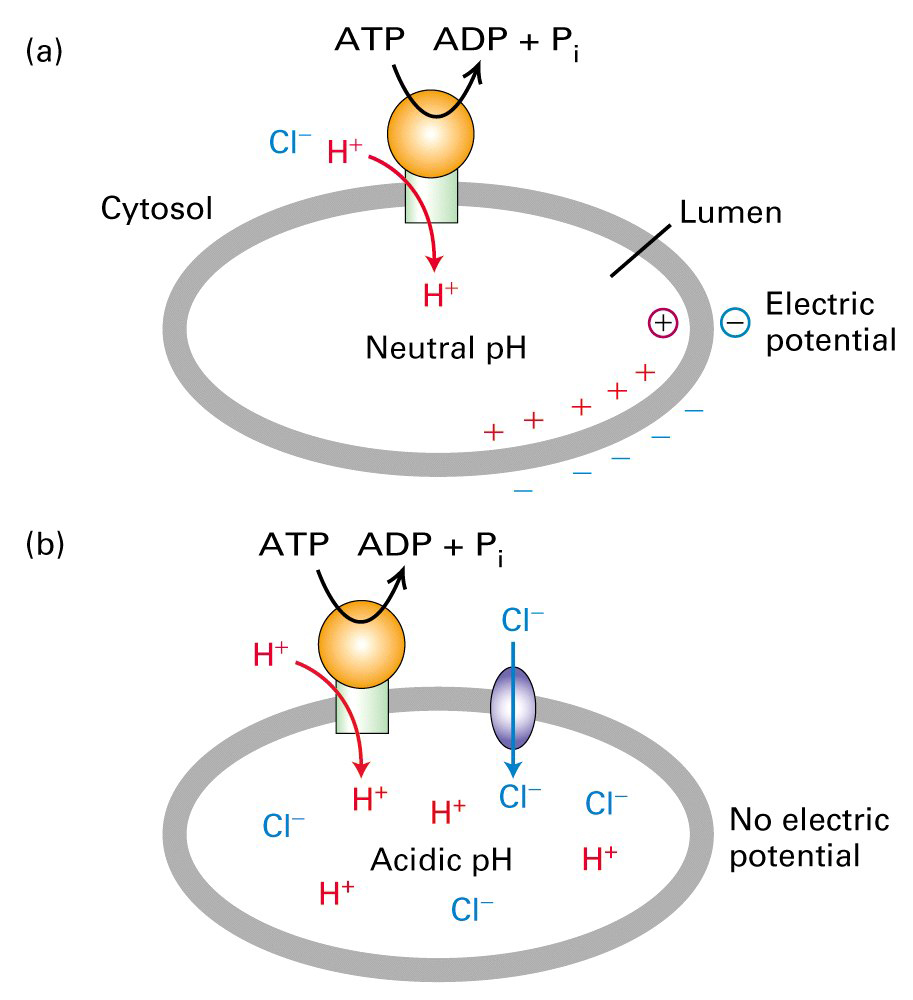
As H+ is transported into the lysosome Cl- flows in to keep a balance. If Cl-
doesn't flow in then there is rapid build up of potential (charge) across the
membrane which would block the further transport of H+. This would occur long
before the lumen becomes acidic because not that many ions need to be
transported to produce the voltage potential.
Other pumps like the H+/K+ ATPase pump (a P-class pump) transport H+ out and
at the same time K+ in to make sure there is no voltage potential created.